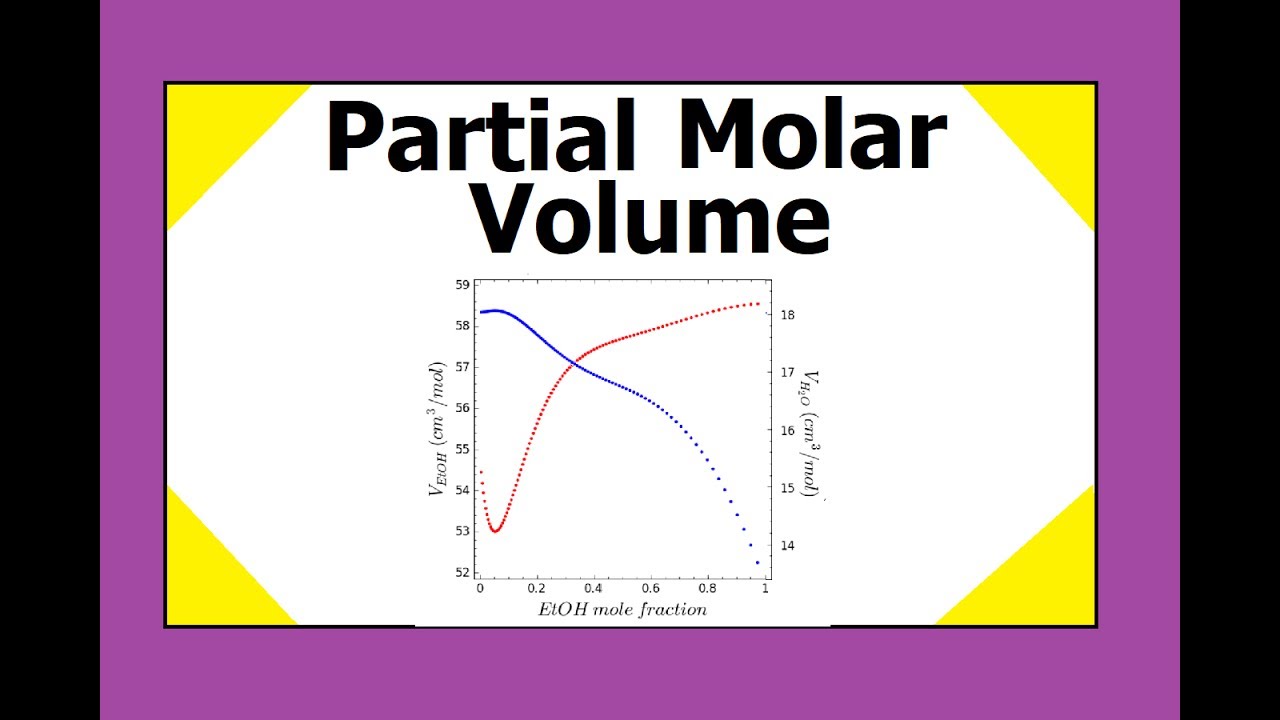
A walk through of an example problem finding an expression for the partial molar volume of a species in a binary mixture using the Gibbs Duhem equation:
The partial molar volume of benzene (1) in cyclohexane (2) at 30°C is given by the following expression:
V ̅1 = 92.6 - 5.28x1 + 2.64x12 [cm3/mol]
Find an expression for the partial molar volume of cyclohexane. The density of cyclohexane at 30°C is 0.768 [g/cm3].
Purdue University Omega Chi Epsilon
Text: Engineering and Chemical Thermodynamics by Koretsky 2nd edition
----------------------------------------------------------------------------------------------------------
Topics: partial molar properties, partial molar volume, binary mixture, pure species volume, infinite dilution, Thermodynamic relations, example problem, fixed temperature, chemical thermodynamics, chemical engineering, thermodynamic properties, how to, engineering, Purdue University, chemical engineering, Purdue university chemical engineering, gibbs duhem equation, CHE 211, Introduction to thermodynamics, partial molar properties, partial molar volume, binary mixture, pure species volume, infinite dilution, Thermodynamic relations, example problem, fixed temperature, chemical thermodynamics, chemical engineering, thermodynamic properties, how to, engineering, Purdue University, chemical engineering, Purdue university chemical engineering, gibbs duhem equation, CHE 211, Introduction to thermodynamics
The partial molar volume of benzene (1) in cyclohexane (2) at 30°C is given by the following expression:
V ̅1 = 92.6 - 5.28x1 + 2.64x12 [cm3/mol]
Find an expression for the partial molar volume of cyclohexane. The density of cyclohexane at 30°C is 0.768 [g/cm3].
The partial molar volume of benzene (1) in cyclohexane (2) at 30°C is given by the following expression:
V ̅1 = 92.6 - 5.28x1 + 2.64x12 [cm3/mol]
Find an expression for the partial molar volume of cyclohexane. The density of cyclohexane at 30°C is 0.768 [g/cm3].
The partial molar volume of benzene (1) in cyclohexane (2) at 30°C is given by the following expression:
V ̅1 = 92.6 - 5.28x1 + 2.64x12 [cm3/mol]
Find an expression for the partial molar volume of cyclohexane. The density of cyclohexane at 30°C is 0.768 [g/cm3].
The partial molar volume of benzene (1) in cyclohexane (2) at 30°C is given by the following expression:
V ̅1 = 92.6 - 5.28x1 + 2.64x12 [cm3/mol]
Find an expression for the partial molar volume of cyclohexane. The density of cyclohexane at 30°C is 0.768 [g/cm3].
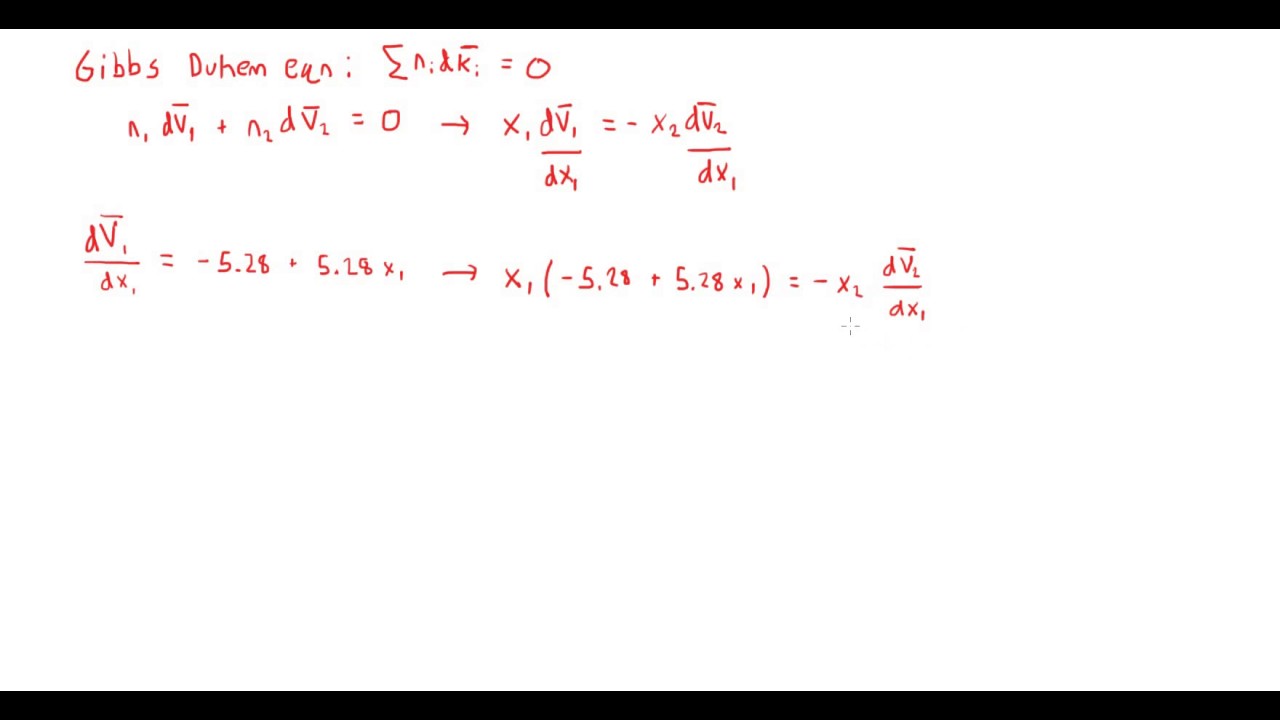
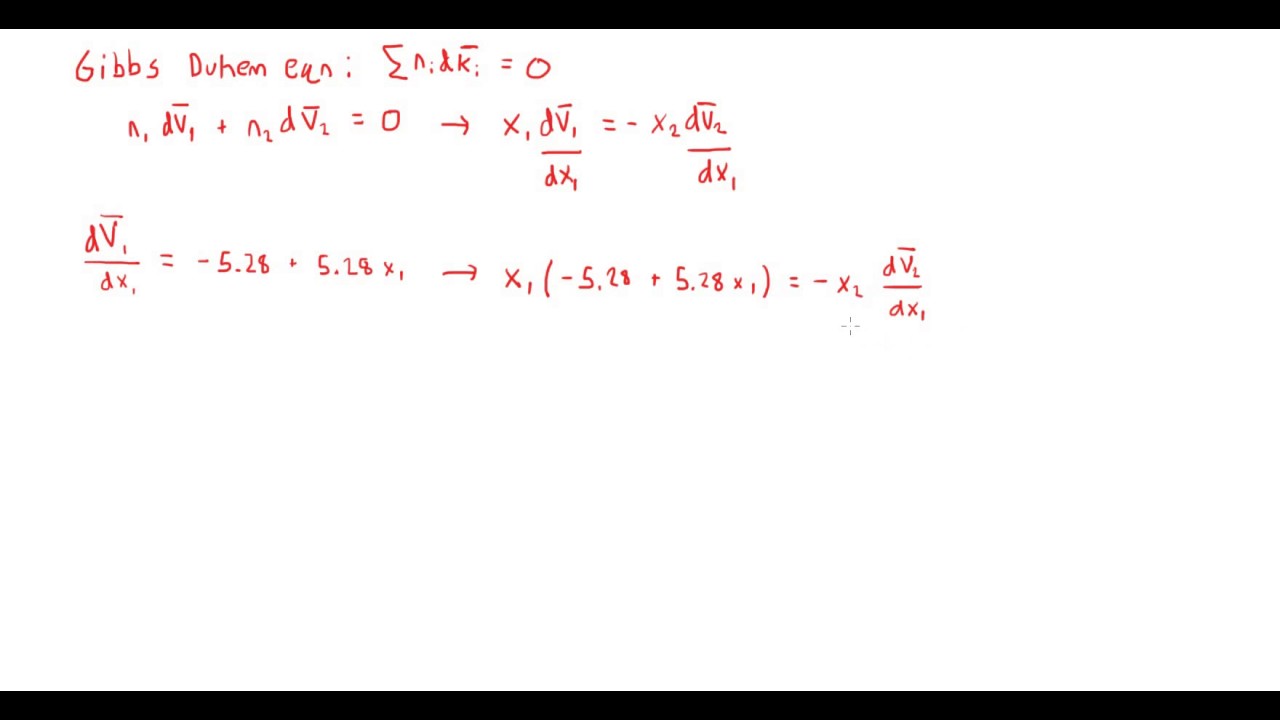


















Информация по комментариям в разработке