🚀 PINACOL–PINACOLONE REARRANGEMENT | 1,2 SHIFT | CARBOCATION REARRANGEMENT
Chemistry for IIT JEE, NEET, GATE, CSIR NET, UGC NET, TIFR, CUTE, SET, TRB
💥 WHY THIS REACTION IS EXAM GOLD? 😎
Because it teaches:
Carbocation stability
1,2 alkyl shift
Acid catalyzed rearrangement
Migration aptitude
Carbon skeleton rearrangement
Very frequently asked in JEE Advanced and GATE 🔥
🧠 WHAT IS PINACOL–PINACOLONE REARRANGEMENT?
Pinacol rearrangement converts:
Vicinal diol
Into
Ketone
Under acidic conditions.
Classic example:
Pinacol → Pinacolone.
⚡ BASIC CONDITIONS
Strong acid such as sulfuric acid
Heat
Protonation of one hydroxyl group
Water leaves
Carbocation formed
🔬 STEP BY STEP MECHANISM
Step 1
One OH group gets protonated.
Step 2
Water leaves.
Carbocation forms.
Step 3
1,2 migration of alkyl group.
Step 4
Rearranged carbocation forms.
Step 5
Deprotonation gives ketone.
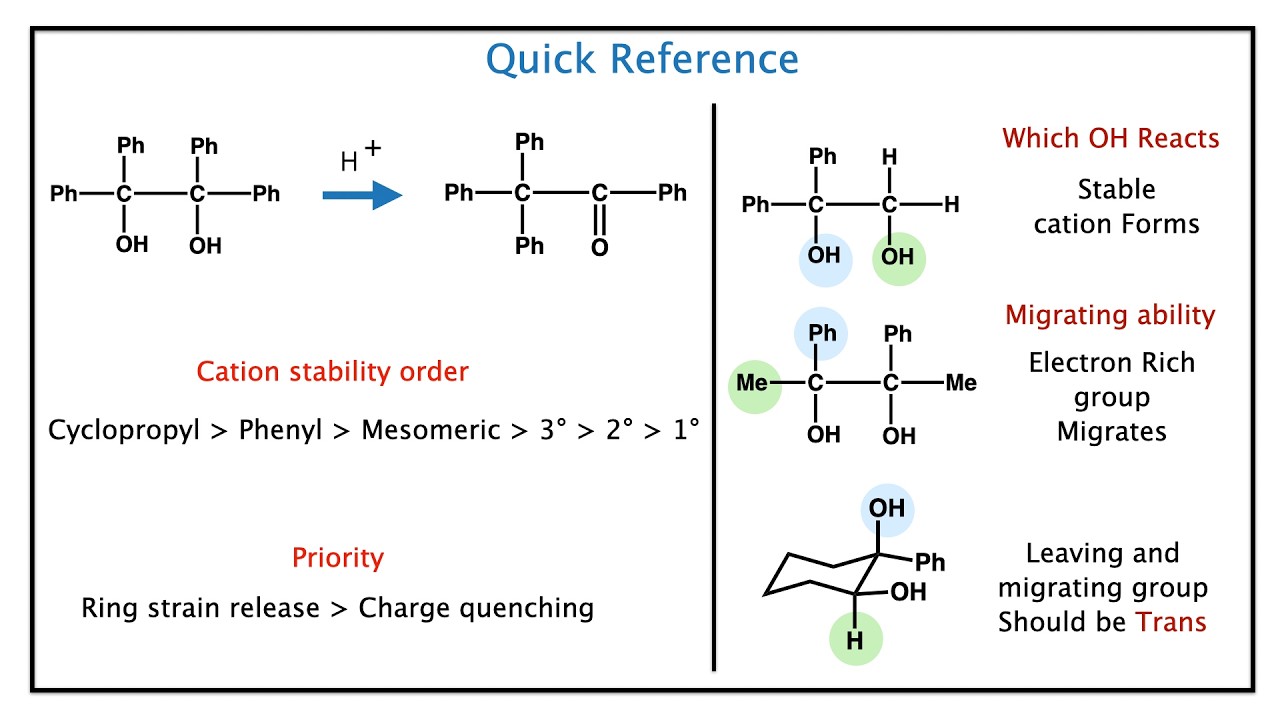
🔥 CORE CONCEPT
Carbocation rearrangement.
Migration occurs to stabilize positive charge.
More stable carbocation favored.
Migration leads to ketone formation.
🎯 WHAT IS MIGRATION APTITUDE?
Which group migrates first?
Hydride shift strongest.
Then tertiary alkyl.
Then secondary.
Then primary.
Aryl groups migrate well due to resonance.
Examiners love asking this order.
😎 SIMPLE ANALOGY
Imagine chair swap in classroom.
Student from stable row moves to unstable seat.
System becomes stable.
That is migration.
📊 IMPORTANT FACTORS
Carbocation stability
Hyperconjugation
Resonance
Steric effects
Nature of migrating group
🎓 EXAM RELEVANCE
Very common in:
JEE Advanced named reactions
NEET carbocation stability
GATE rearrangement mechanism
CSIR NET migration aptitude
TIFR advanced organic
Typical questions:
Predict product
Identify migrating group
Compare stability
Determine major ketone
🔥 EXAM SHORTCUTS
🔥 Vicinal diol + acid
🔥 Protonation then water loss
🔥 Carbocation formed
🔥 1,2 shift occurs
🔥 More stable carbocation favored
🔥 Final product is ketone
❌ COMMON MISTAKES
Forgetting protonation step
Wrong migration order
Ignoring carbocation stability
Confusing with benzilic acid rearrangement
📌 QUICK REVISION NOTES
Acid catalyzed reaction
Water loss
Carbocation intermediate
1,2 alkyl or hydride shift
Ketone formation
Migration aptitude important
📝 SHORT NOTES FOR BOARDS
Define pinacol rearrangement
Mention acid condition
Explain carbocation formation
Describe 1,2 shift
State ketone formation
🚀 ADVANCED INSIGHT
Pinacol rearrangement demonstrates:
Carbocation driven rearrangement
Stability control
Migration aptitude concept
Structural reorganization
It builds strong foundation for understanding Wagner Meerwein rearrangement and other carbocation rearrangements.
Understanding migration logic improves product prediction accuracy.
📌 SUMMARY OF DESCRIPTION
Pinacol–pinacolone rearrangement converts vicinal diol into ketone under acidic conditions.
Water leaves forming carbocation.
1,2 migration stabilizes positive charge.
Final product is ketone.
Mastering migration aptitude and carbocation stability is essential for solving advanced rearrangement questions in competitive exams 💯🔥
🔑 30 IMPORTANT KEYWORDS
Pinacol rearrangement, Pinacolone formation, Vicinal diol reaction, Carbocation rearrangement, 1,2 shift, Migration aptitude, Acid catalyzed reaction, Organic chemistry, JEE Advanced chemistry, NEET organic chemistry, GATE chemistry, CSIR NET chemistry, TIFR entrance exam, Named reaction, Reaction mechanism, Competitive exam chemistry, Product prediction, Concept clarity, Hyperconjugation, Resonance stabilization, Chemistry revision, Exam trick, Advanced organic chemistry, Alkyl shift, Hydride shift, Ketone formation, Structural rearrangement, Mechanism mastery, Migration order, Competitive exams 🚀
📌 30 HASHTAGS
#PinacolRearrangement
#Pinacolone
#Carbocation
#OrganicChemistry
#JEE2026
#NEET2026
#IITJEE
#JEEMain
#JEEAdvanced
#NEETPreparation
#GATEChemistry
#CSIRNET
#UGCNET
#TIFR
#SETExam
#TRBExam
#CUTEExam
#ChemistryTricks
#Class12Chemistry
#ReactionMechanism
#ChemistryRevision
#CompetitiveExams
#ChemistryMadeEasy
#MechanismMaster
#ExamPreparation
#AdvancedChemistry
#ConceptClarity
#MigrationAptitude
#RearrangementReaction
#OrganicSynthesis



















Информация по комментариям в разработке